Infertility treatment with ART
ART procedures for a successful pregnancy

BAP/ART procedures
BAP is an abbreviation for Biomedically Assisted Procreation. ART is an abbreviation for Assisted Reproductive Technology. Both abbrevations have the same practical meaning.
ART is an abbreviation for Assisted Reproductive Technology.
ART procedures are fertility procedures that use biomedical science to achieve pregnancy in ways other than sexual intercourse. All ART procedures are carried out in accordance with the achievements and development of biomedical science (Article 4 of the ZZNPOBMP).
Louise Brown, the first child conceived using ART, was born on July 25, 1979, in Manchester, Great Britain.

Intracorporeal fertilisation:
- Introduction of sperm cells into the reproductive organs of a woman (intrauterine insemination or IUI).
Extracorporeal fertilisation:
- Oocyte and sperm cell fusion outside of the female body (IVF, ICSI).
- Introduction of early embryos into the reproductive organs of a woman (embryo transfer or ET).
Intrauterine Insemination (IUI)
One of the most basic methods of artificial insemination is intrauterine insemination (IUI). It is the first treatment option for couples who have been unable to conceive using other fertility treatments.
This is especially true for couples who fail due to the male partner’s sperm being of slightly lower quality or the female partner’s irregular ovulation. This program is also appropriate for patients who have an infertility immune factor.
The IUI method facilitates the passage of sperm to the egg. Sperms avoid the potentially harmful effects of the vaginal and cervix environments. The time required to reach the egg is reduced.
The IUI procedure:
- When preparing the partner’s seminal ejaculation in the IVF laboratory, a sample with the greatest number of quality sperm possible is obtained.
- Using a long thin catheter (1.8 mm diameter), the appropriate dose of seminal fluid is introduced directly into the woman’s uterus.
Before the IUI procedure, a woman’s fallopian tube passage test must be performed. A partner’s fresh seminal ejaculation is typically used. At the same time, mild hormonal stimulation of the female partner’s ovulation is usually performed. The goal of stimulation prior to IUI is to mature 1 to 2 follicles. Stimulation increases the chances of becoming pregnant. The procedure is completely painless.
During the first cycle of IUI treatment, 5 to 10% of women become pregnant. Success is determined by the woman’s age and decreases with age. It is almost never done after the age of 40. If you are unable to conceive after 3 to 4 cycles of IUI, your doctor will recommend a more effective method – in vitro fertilisation (IVF, ICSI).
Extracorporeal fertilisation or in vitro fertilisation (IVF, ICSI)
Extracorporeal fertilisation or in vitro fertilisation (IVF, ICSI) is a method of symptomatic treatment in which a woman’s oocytes are fertilised with a partner’s sperm outside the female body – in vitro fertilisation.
The IVF/ICSI procedure begins with the hormonal stimulation of a woman, which is required to obtain more eggs than during the normal ovulation cycle. A doctor monitors the course of stimulation using an ultrasound. The mature eggs are collected by puncturing the follicles under ultrasound control. The partner’s sperm must also be collected on the day the eggs are collected. The resulting eggs are then fertilized with the partner’s sperm, and the embryos are grown outside of the female body in an incubator. The best embryo is transferred to the woman’s womb after a few days of cultivation (maximum 6 days) – the embryo transfer (ET). The remaining embryos are frozen.
Traditional IVF
In the case of IVF, two basic methods are distinguished. If the sperm is of good quality, traditional IVF is less invasive and more successful. In this case, the oocytes are incubated with the sperm for 12 to 24 hours.
IVF-ICSI
If the sperm quality is poor, only ICSI (Intracytoplasmic sperm injection) is considered. The basic idea behind this method is to inject healthy sperm directly into the cytoplasm of a mature egg. Although the insemination rate is higher than with traditional IVF, ICSI is a fairly invasive procedure that requires an experienced embryologist, and the insemination rate is slightly lower.

Cultivation of zygotes and embryos
Fertilised oocytes or zygotes are extremely sensitive to any deviation from ideal conditions, such as those found in the uterus. To that end, cultivators and incubators for growing early embryos have emerged alongside the advancement of modern assisted reproduction methods. For the embryo to develop successfully outside the uterus, all parameters (pH, temperature, humidity, chemical composition of the environment) must be as close to those found in the fallopian tube or uterus as possible. The overall development of the embryo in the laboratory must mimic changes in the conditions of the fertilised oocyte’s travel through the fallopian tube and the uterus. As a result, all procedures for handling embryos in the laboratory are carefully planned and monitored.
Transfer of the embryo to the uterus
Embryos can develop outside the body for up to 5-6 days, or until they are transferred. The embryos grow in an incubator under conditions similar to those found in the fallopian tube or uterus.
The gynaecologist, embryologist, and couple discuss several criteria on the day of transmission:
- number of embryos;
- the quality of embryos;
- anamnesis data;
- the couple’s wishes.
Typically, only 1 or 2 of the best embryos are transferred into the uterus.
The decision is made by the embryologist, who evaluates and monitors the embryos from the puncture to the transfer to the uterus. Today, embryologists can benefit from time-lapse technology to avoid the subjective factor of this choice. These are incubators with built-in cameras that continuously record the embryo’s development. The morphometric data obtained on each embryo is then compared with the embryos in the system for which we know their fate (collapse, nesting, childbirth), and the computer proposes which embryo has the best potential using certain algorithms.

ART/BAP laboratory procedures

Oocyte identification and isolation
The embryologist collects follicular fluid and identifies the oocytes in it during ovarian puncture. Because oocytes are extremely sensitive to temperature changes, it is critical that this process be completed as quickly as possible. The follicular fluid is collected in heated tubes that are then collected in a similarly heated thermal block, reducing the possibility of temperature fluctuations.
The embryologist then looks for oocytes surrounded by granulosa cells on the heated surface of a dust-free chamber. The entire complex resembles a cloud and can be seen with the naked eye.
The cells are collected and transferred to a suitable medium containing HEPES buffer, allowing stable conditions outside the incubator. Following that, the cells are transferred to a pre-prepared medium and placed in an incubator until fertilisation.
Traditional IVF
As previously stated, traditional IVF is the closest thing to the natural fertilisation of an egg in a woman’s body. A purified and concentrated sperm of the partner is added to the oocyte-granulosa cell complexes in a petri dish, allowing the fertile sperm to fertilise the oocyte. The method is only effective if the sperm is of high quality. The embryologist removes the granulosa cells the next day and checks for fertilisation. The embryologist also excretes all irregularly fertilised cells (triploid and monoploid), which occur when the egg is fertilised by two sperms or when pronuclei form irregularly during the chromosome fusion process. Because embryos are generally chromosomally irregular, we can distinguish such zygotes.

IVF-ICSI
The direct introduction of sperm into the oocyte is without a doubt a milestone in reproductive medicine and the treatment of infertility. Couples who were previously forced to seek a solution through adoption or donated cells now have the option of having a child with their own cells.
The first successful ICSI procedure was carried out in 1991 by the Italian embryologist Dr Palermo at the University of Brussels. Since then, nearly 5,000,000 children have been born using this method worldwide, and ICSI is now a routine procedure that allows us to successfully treat even the most difficult forms of male infertility.
The procedure for the acquisition and collection of oocytes is identical to the traditional IVF procedure. However, because ICSI involves oocyte manipulation, the granulosa sheath must be removed prior to the procedure. The embryologist carefully removes the granulosa cells with a thin pipette immediately before introducing the sperm, allowing the oocyte to attach to the so-called holding pipette. Only oocytes in the second metaphase of the meiotic division (MII) are used, which means that they are capable of continuing meiosis, chromosome aggregation, and zygote formation during this brief time window. The embryologist searches for morphologically suitable sperm with the second input pipette and sucks it into the pipette using a micromanipulator device. The powerful microscope magnification and micrometric movements of the micromanipulator pipettes allow us to introduce sperm directly into the cytoplasm with as little damage to the oocyte as possible.
The procedure, like traditional IVF, is evaluated the following day with the confirmation of diploid zygotes.
Cultivation of embryos in an incubator
What happens 24 hours after insemination?
- The embryos are transferred to a fresh, pre-calibrated medium;
- The performance of fertilisation is assessed;
- Unfertilised and improperly fertilised egg cells are separated from the rest.
The embryos are then left undisturbed in the incubator for up to 72 hours after fertilisation, when their development is monitored again.
If we decide to transfer embryos on day 3, we do so.
If the cultivation period is extended until day 5, the embryos are transferred to a medium that allows the embryo to develop to the blastocyst stage, which can take up to 5 days.
Cultivation mediums
In our ZCD centre, we use Vitrolife prepared mediums. The complete product line allows us to provide a stable and controlled environment for embryos. All mediums are manufactured in accordance with the most stringent quality and transportation control standards. In addition, we perform strict quality control and internal control of all procedures and medium manipulation in our laboratory. To ensure a safe environment for embryos, we regularly inspect refrigerators, incubators, and pH mediums.
Freezing of embryos
The procedures for freezing and feeding embryos significantly improve the overall performance of ART procedures. Supernumerary embryos of good quality collected via ICSI or IVF are frozen using modern techniques and stored in liquid nitrogen for an extended period of time. Traditional gradual freezing has largely replaced so-called ultra-short freezing or vitrification in recent years. The process is short and extremely efficient.
Special solutions are used to dehydrate the embryos, preventing the formation of water crystals and subsequent embryo damage. By the addition of cryoprotectants, they are immersed directly in liquid nitrogen at a temperature of -196ºC.
More than 90% of the embryos survive, and the birth rate is comparable to that of fresh embryos.
Additional ART laboratory procedures
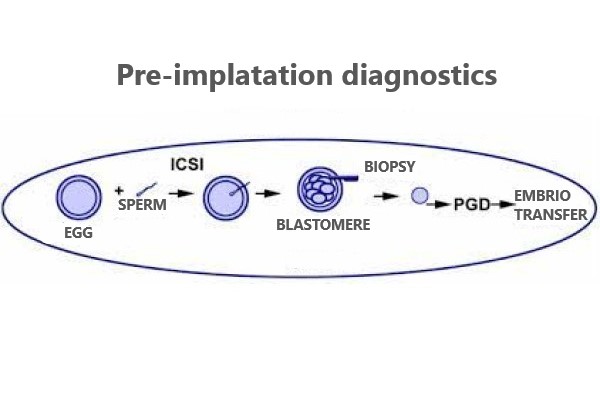
Pre-implantation diagnostics
Chromosomal and genetic defects are common causes of infertility treatment failure. Certain mutations are also passed down through generations. Despite cutting-edge extracorporeal fertilisation techniques, implantation and development of a chromosomally or genetically irregular embryo are impossible. Defects can be inherited, occur during gametogenesis, or occur after fertilisation. Fortunately, methods are now available to detect these abnormalities even before the embryo is transferred to the uterus or before biomedical fertilisation. By PCR, FISH, or NGS procedures, a biopsy of a part of the embryo (blastomere, trophectoderm, or polar body) reveals such specific monogenic or chromosomal mutations. In this way, only healthy embryos are transferred into the uterus. In addition to a fairly invasive embryo biopsy, some recent methods allow non-invasive testing of culture medium in which embryos are developed in incubators, and these methods exploit the so-called free DNA found in these media.

Prolonged embryo cultivation, or blastocyst transmission
We can successfully grow a larger number of embryos thanks to high-quality media, stable incubators, and new ovarian stimulation protocols. In such cases, we usually decide to extend the cultivation, which means that the embryos are grown in the incubator until they reach the blastocyst stage on the fifth day. This procedure allows us to better select embryos and is also more appropriate in terms of embryo level synchronicity with the endometrium.
Laser-assisted hatching
When multiple ART procedures fail, especially when the embryos are of high quality, we decide to artificially thin or terminate the embryo’s envelope to facilitate implantation (nesting) in the uterus. This is also decided in the case of an overly thick envelope or the transfer of frozen embryos.

EmbryoGlue
Vitrolife has also developed a special medium that is used exclusively for embryo transfer. Because this is a critical part of the ART process, the medium in which it is done is also important for an optimal result. EmbryoGlue (EG) is not an adhesive, but it contains a high proportion of hyaluronan and recombinant human albumin, which mimics the microenvironment in the uterus where the embryo is implanted. Numerous studies have confirmed EmbryoGlue’s added value in ART procedures, which increases the likelihood of having a baby.
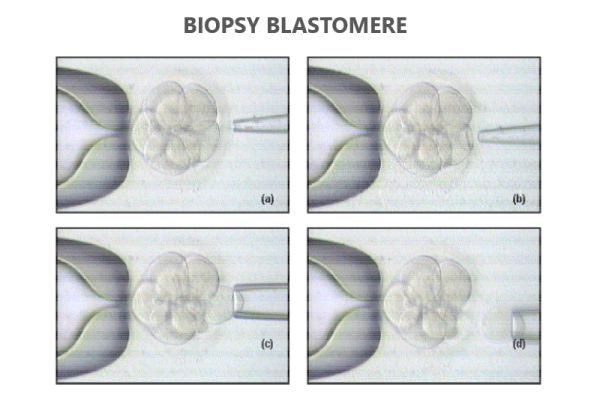
Biopsy blastomere
A biopsy blastomere is an invasive procedure that involves taking one or more cells through a small opening in the embryo’s envelope in order to perform genetic or chromosomal analysis on the developing embryo. Following proper fixation and preparation of the harvested cells, they are sent to a genetic laboratory, where they can determine whether the embryo carries the gene for a hereditary disease or whether its number and chromosome structure are correct based on certain tests.
BAP success rate

BAP success rate
For many couples with fertility problems, very simple treatments can help. We always proceed from the least complex treatment to the standard IVF protocols.
The general success rate of IVF procedures is 25–30% per procedure.
It should be borne in mind that in women over 37 years of age, the success rate of this method decreases due to the poorer quality of the eggs.
Pregnancy test after BAP
Take a rapid pregnancy test two weeks after embryo transfer. Use the first morning urine. Pregnancy can be further confirmed by taking a blood sample and determining the value of beta hCG. If the test is positive, it is a biochemical pregnancy.
If the pregnancy test is positive, you should continue with your prescribed treatment and make an appointment for a pregnancy ultrasound examination. The first ultrasound examination is carried out at the earliest four (4) weeks after the transfer of the embryos. This examination is used to determine where the pregnancy is, how many amniotic sacs are present, whether it is growing and whether the pregnancy is viable (with the presence of heartbeats).
If your biochemical pregnancy test is negative, do not despair, but contact our centre. We will discuss further diagnostics and treatment with you.
